-
News+GNTbm's self-developed anti-cancer new drug GNTbm-38 has obtained IND approval from the U.S. FDA for Phase I clinical trials
2026-03-05 -
About Us+
Great Novel Therapeutics Biotech & Medicals Corporation (GNTbm) is a clinical-stage biopharmaceutical company dedicated to the research and development of innovative therapies for advanced and refractory cancers. Building upon its expertise in epigenetic modulators, GNTbm has strategically expanded into the high-potential field of cancer immunotherapy.The company’s proprietary R&D pipeline features next-generation epigenetic immunoactivators and immunoregulatory multi-tyrosine kinase inhibitors (multi-TKIs). These agents serve as backbone therapies in next-generation cancer immunotherapy, specifically designed to modulate the tumor microenvironment (TME). By developing broad-spectrum, orally bioavailable immunotherapies, GNTbm aims to address the significant unmet medical needs of patients with advanced and refractory malignancies worldwide.
-
Immuno-Oncology therapy+
Unlike conventional chemotherapy or radiotherapy, cancer immunotherapy does not target malignant cells directly. Instead, it functions by reawakening or bolstering the patient’s own immune system to restore its anti-tumor efficacy and establish immunological memory, thereby preventing recurrence. Among various approaches, tumor microenvironment (TME) modulators hold the greatest potential as key regulators of the immune response. By dismantling the immunosuppressive barriers within the tumor, these agents transform 'cold tumors' into targetable 'hot tumors,' fundamentally redefining the ecological landscape of the anti-cancer battlefield.
-
Research Highlights+
Kepida® (Tucidinostat/Chidamide) is a subtype-selective histone deacetylase (HDAC) inhibitor with a novel chemical structure and global patent protection. It achieves its anti-tumor epigenetic regulatory mechanism by selectively inhibiting Class I HDAC isoforms 1, 2, and 3, classifying it as a next-generation epigenetic modulator.
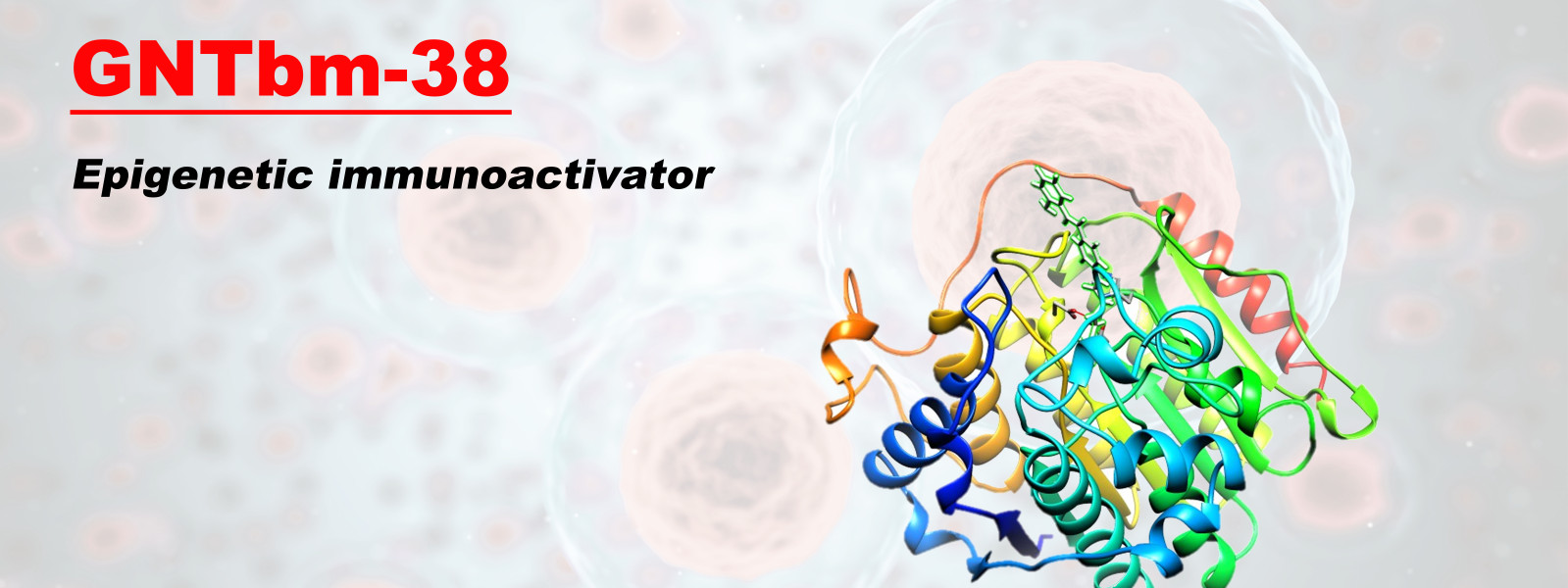
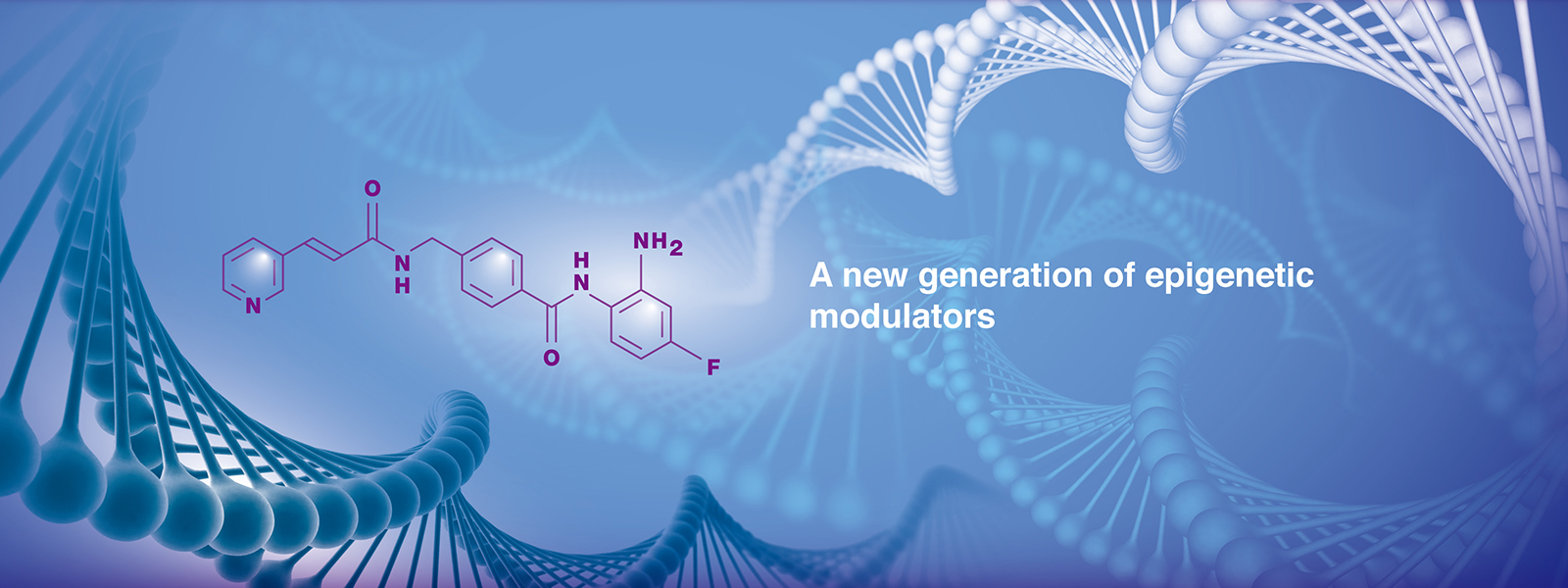
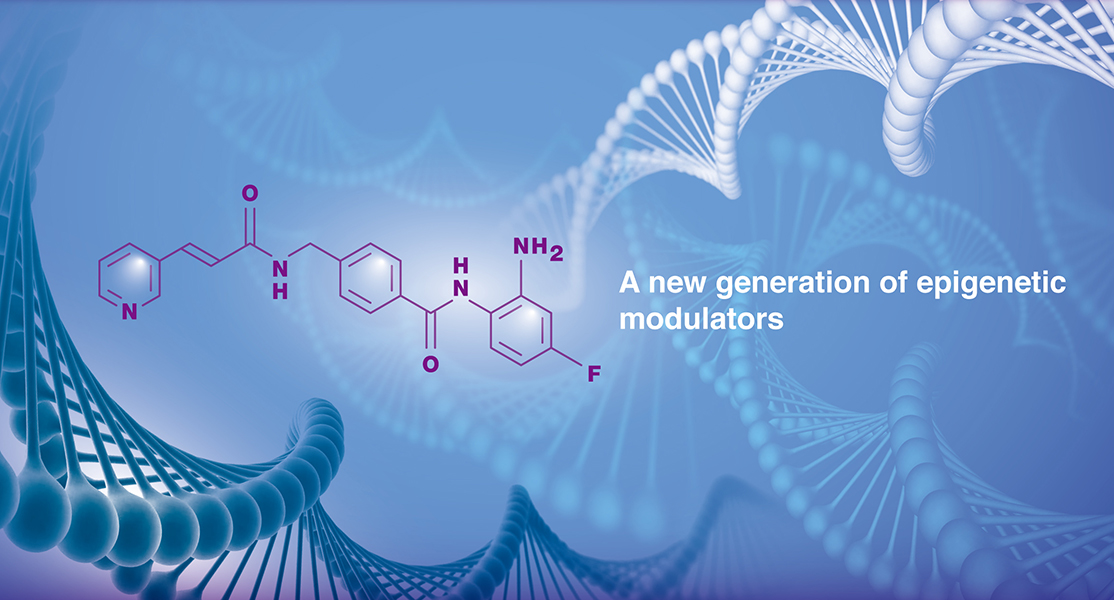
GNTbm-38 is a proprietary, orally bioavailable small molecule New Chemical Entity (NCE) developed by GNT Biotech Hub. As a next-generation immuno-epigenetic activator, it is currently undergoing clinical research, with plans to submit Investigational New Drug (IND) applications in the United States, Taiwan, and China.
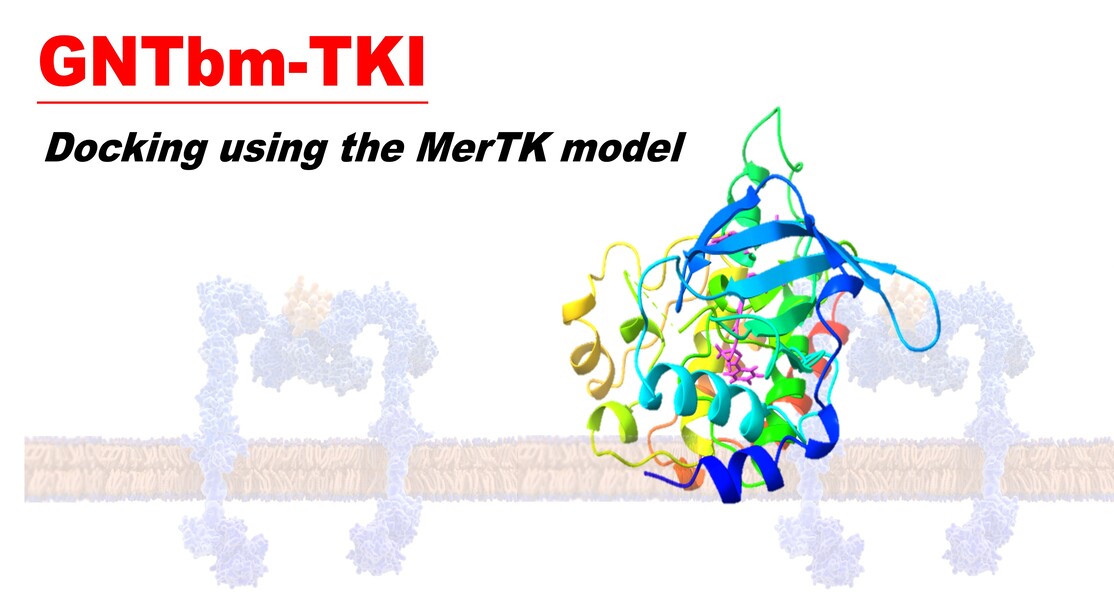
GNTbm-TKI is a potent immuno-modulatory multi-kinase inhibitor designed for the treatment of various solid tumors. Currently in the pre-clinical stage, it is projected to file for IND by the end of 2026.