Potent immune-regulatory multi-tyrosine kinase inhibitor
In the past 20 years, multiple tyrosine kinase inhibitors (TKIs) have been approved for the treatment of various cancers and other indications. However, most existing TKIs primarily exert their effects through direct inhibition of tumor growth or angiogenesis, with limited ability to modulate the tumor immune microenvironment (TME).
In response, the R&D team at GNTbm has focused on developing a next-generation multi-kinase inhibitor with potent immune-modulatory activity, resulting in the success of GNTbm-TKI, a structurally novel immune-regulatory TKI designed for application in cancer immunotherapy.
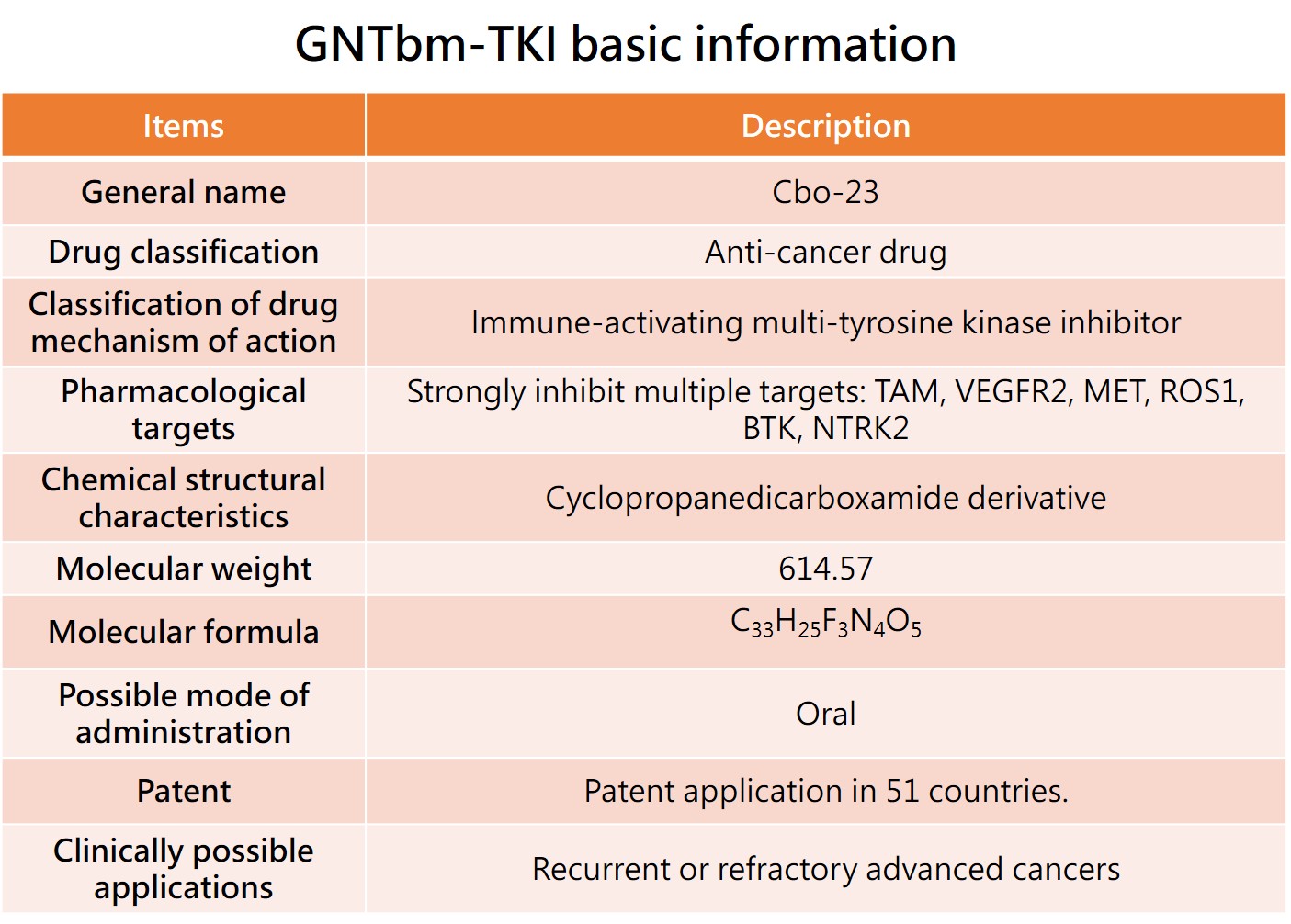
GNTbm-TKI is an innovative orally administered small-molecule candidate identified through years of research and screening, demonstrating robust immune-regulatory activity. Preclinical studies—including enzymatic, cellular, and in vivo models—have shown that GNTbm-TKI can modulate multiple immune-related signaling pathways by concurrently targeting key kinases involved in tumor immune evasion and immunosuppression, including TYRO-3, AXL, c-MER (TAM family), BTK, ROS1, NTRK2, VEGFR2, and MET. This multi-target design not only inhibits tumor growth and angiogenesis but also effectively remodels the immunosuppressive TME.
Further studies indicate that GNTbm-TKI exhibits significant synergy when combined with HDAC inhibitors (e.g., GNTbm-38). Through a dual mechanism of “epigenetic modulation × kinase signaling regulation,” this combination can convert immune-cold tumors into immune-hot tumors, markedly enhancing anti-tumor immune responses. This property positions GNTbm-TKI not only as an active monotherapy (e.g., for advanced neuroendocrine tumors) but also as a backbone therapy in combination regimens.
Compared to conventional TKIs, GNTbm-TKI’s competitive advantage lies in its immune-directed multi-target design, which can simultaneously impact tumor cells, tumor vasculature, and immune cells (e.g., macrophages and dendritic cells), thereby potentiating the efficacy of immune checkpoint inhibitors. Accordingly, GNTbm-TKI can be flexibly applied in multiple combination strategies, including:
(1) Combination with HDAC inhibitors (e.g., Tucidinostat or GNTbm-38)(2) Combination with immune checkpoint inhibitors (anti-PD-1/PD-L1)
(3) Combination with anti-PD-1/VEGF bispecific antibodies
Through these strategies, GNTbm-TKI is expected to become a key agent in cancer immunotherapy, with broad applicability across various advanced solid tumors.
GNTbm-TKI is currently in the preclinical stage, with Phase I clinical trials projected to commence in 2027. Subsequent development will prioritize combination studies with GNTbm-38, progressing to Phase II clinical trials, with orphan indications serving as initial entry points. Accelerated approval is expected via single-arm pivotal Phase II studies.
The overall development strategy centers on “GNTbm-TKI + I (Immunotherapy),” where I encompasses HDAC inhibitors, immune checkpoint inhibitors, and bispecific antibodies. Leveraging its potent immune-modulatory capacity, GNTbm-TKI is poised to overcome immune-cold tumor limitations across multiple solid tumors and establish a scalable platform for tumor immunotherapy.
1. The first indication of GNTbm-TKI --- Neuroendocrine tumors
Neuroendocrine tumors (NETs) are a type of rare tumor that commonly occur in the neuroendocrine cells of the human body. These cells possess characteristics of both nerve cells and hormone-secreting endocrine cells. Most neuroendocrine tumors are malignant. NETs most commonly occur in the gastrointestinal tract, with approximately 20% occurring in the colon, 19% in the small intestine, and 4% in the appendix. The second most common site for NETs is the lungs, with about 30% of cases possibly occurring in the bronchial system. NETs of the gastrointestinal tract and lungs are both referred to as carcinoids.
In addition, approximately 7% of NETs occur in the pancreas, a condition known as pancreatic NET (pNET) or pancreatic islet cell tumor. Common symptoms of NETs or islet cell tumors include unexplained weight gain or loss, abdominal pain, vomiting blood, anxiety, sweating, headaches, loss of consciousness, inflammation of the tongue and mouth, abdominal lumps or masses, seizures, blurred vision, rapid heart rate, and jaundice.
According to statistics from Datamonitor Healthcare, the number of global NET cases in 2024 is approximately 588,200, and it is projected to increase to 679,400 by 2031. It is estimated that Asia will have the highest number of NET cases in 2024 (357,600 cases). According to Global Market Insights report, the incidence of NETs continues to rise in North America, Asia, and Europe, with the most significant increase observed in North America. For example, in the United States, the incidence of NETs has grown more than sixfold over the past 40 years, with the highest incidence rate in localized NET tumors.
In April 2025, Cabozantinib was approved by the U.S. FDA for the treatment of locally advanced or metastatic, previously treated, unresectable well-differentiated pancreatic neuroendocrine tumors (pNETs) and extra-pancreatic neuroendocrine tumors (epNETs). The targets inhibited by Cabozantinib are similar to those of Zanzalintinib and GNTbm-TKI (TAM, VEGFR2, c-MET), primarily inhibiting tumor growth and development, tumor invasion and metastasis, tumor angiogenesis, and immune regulatory activity. Compared to Cabozantinib and Zanzalintinib, the main distinction of GNTbm-TKI is its stronger inhibition of ROS1, BTK, NRTK2, and Tie2, along with more pronounced immune activation.
Therefore, GNTbm-TKI not only has potential as a monotherapy for neuroendocrine tumors (NETs) but also, through its potent immune-activating capability, holds promise for combination with epigenetic modulators (e.g., GNTbm-38) or immunotherapies. This approach enables the development of differentiated tumor immunotherapy strategies and the potential expansion into multiple advanced solid tumor indications.
2. The development of GNTbm-TKI's new indications adopt a basket trial strategy --- advanced kidney cancer, advanced head and neck cancer, advanced colorectal cancer
Basket Trials represent a breakthrough in precision medicine by transcending traditional organ-based classifications. The core logic is "treating different diseases with a common mechanism": patients with various malignancies sharing the same genetic mutations or biomarkers are enrolled in a single "basket" to receive a specific targeted therapy. This design efficiently validates a drug’s broad-spectrum efficacy and accelerates Proof-of-Concept (POC), providing a high-efficiency pathway for developing treatments against refractory cancers with specific epigenetic signatures.
The triplet combination of GNTbm-TKI, GNTbm-38, and an anti-PD-1/VEGF bispecific antibody represents a highly promising, next-generation cancer immunotherapy strategy. By integrating kinase signaling modulation, epigenetic reprogramming, and dual immune checkpoint/angiogenesis blockade, this combination synergistically targets both tumor cells and the tumor microenvironment (TME) to achieve comprehensive anti-tumor effects.
Preclinical studies have confirmed that the doublet of GNTbm-TKI and GNTbm-38 significantly remodels the TME and activates immune responses, demonstrating superior anti-tumor activity. Furthermore, the combination of GNTbm-38 with an anti-PD-1 antibody has validated robust tumor inhibition. Building on these results, the integration into a triplet regimen (GNTbm-TKI + GNTbm-38 + anti-PD-1) has shown enhanced immunoactivation and anticancer efficacy, supporting its profound synergistic potential.
From a strategic perspective, compared to the conventional doublet of anti-PD-1 and Bevacizumab, the anti-PD-1/VEGF bispecific antibody simultaneously blocks immune checkpoints and angiogenic signaling, offering superior therapeutic integration and a potentially lower treatment burden. Incorporating this bispecific agent into the triplet combination is expected to further enhance efficacy while optimizing the safety profile.
Based on the consolidated mechanism and preclinical evidence, this triplet therapy exhibits potent TME remodeling and immunoactivation capabilities, with broad applicability across various advanced solid tumors.
The initial priority indications include:
(1) Advanced Renal Cell Carcinoma (First-line)
(2) Advanced Head and Neck Cancer (First-line)
(3) Advanced Colorectal Cancer (Second-line)
For the clinical development of this triplet combination, a basket trial design will be employed to simultaneously evaluate efficacy and safety across multiple indications. The initial phase plans to enroll approximately 20 patients per indication to establish Proof-of-Concept (POC).
Upon reaching the predefined primary efficacy endpoints, the program will advance into a pivotal Phase III study. This stage will employ a multiregional clinical trial (MRCT) strategy, spanning the U.S., EU, Canada, Australia, Taiwan, China, and Japan, to systematically validate efficacy and safety, thereby expediting global clinical development and regulatory approval.