Uncovering the anti-cancer mechanisms of Immuno-Oncology
1. Cancer Immunotherapy
Cancer immunotherapy represents a major breakthrough in modern oncology. Cancer cells are particularly challenging to eradicate because they are highly adept at evading immune surveillance. By expressing specific proteins, they can deceive immune cells into recognizing them as normal tissue, thereby escaping immune attack.
Unlike conventional chemotherapy or radiotherapy, which directly target cancer cells, the core principle of cancer immunotherapy is to re-activate or enhance the patient’s immune system, enabling it to recognize and eliminate tumor cells. Currently, three major therapeutic modalities are widely used: immune checkpoint inhibitors, CAR-T cell therapy, and tumor microenvironment (TME) modulators.
The fundamental concept underlying these approaches is the precise modulation of tumors with different immune phenotypes. Tumors can generally be categorized as “immune-hot” or “immune-cold.” Immune-hot tumors are characterized by the presence of tumor-infiltrating T cells; however, these immune cells are often suppressed by inhibitory signals, such as immune checkpoint molecules or immunosuppressive cell populations, which limit their cytotoxic function. In contrast, immune-cold tumors lack sufficient T cell infiltration, often due to a tumor microenvironment that excludes immune cells, making it difficult for the immune system to recognize and initiate an effective anti-tumor response.
Accordingly, different immunotherapeutic strategies are employed. Immune checkpoint inhibitors function by releasing the “brakes” on T cells, thereby restoring pre-existing anti-tumor immunity. CAR-T cell therapy involves engineering a patient’s own T cells to enhance their ability to specifically recognize and attack cancer cells. In contrast, TME modulation focuses on reshaping the tumor microenvironment to facilitate immune cell infiltration, potentially converting immune-cold tumors into more immunologically responsive states and ultimately improving therapeutic outcomes.
2. Tumor Microenvironment Remodeling screening platform
Cancer immunotherapy has become the most promising area with novel mechanisms for the treatment of advanced cancer, mainly by reactivating the patient's immune system, correcting the mechanism of immune escape that had occurred, and releasing the activity of CTLs to attack tumors and generate immune memory.
After 10 years of development, cancer immunotherapy, mainly focused on ICIs, has entered a new generation of cancer immunotherapy. The new approach considers the need of remodeling TME, primarily using anti-PD-1/VEGF or anti-PD-L1/VEGF bispecific antibody (BSA). Although BSAs are regarded as superior to monoclonal antibodies in terms of the efficacy, their ability to enhance anti-angiogenesis is limited, and they do not truly address the challenges in treating cold tumors. The benefits of BSAs in cancer immunotherapy still do not meet the treatment needs of patients with cold tumors. Therefore, it is necessary to combine with other drug X to enhance the remodeling of the TME and achieve better outcomes in cancer immunotherapy.
.jpg)
3. A new generation of epigenetic immunoactivator screening platform
Epigenetics refers to heritable changes in gene expression that occur without altering the underlying DNA sequence. In oncology, epigenetic immunoactivators represent a transformative breakthrough by reprogramming cancer cells to “unmask” previously immune-evasive tumors. By restoring suppressed immune responses and enabling T cells to effectively eliminate malignant cells, this approach offers a paradigm shift beyond conventional chemotherapy—delivering potent anti-tumor activity with the potential for reduced systemic toxicity.
While epigenetic modulators such as histone deacetylase inhibitors (HDAC inhibitors, HDACi) have been developed for over two decades, with multiple agents approved worldwide, most lack meaningful immunoregulatory activity. GNTbm has identified that structural differences fundamentally determine immunoregulatory capacity. This insight highlights a critical industry gap: true next-generation therapies must integrate both epigenetic reprogramming and immune activation to achieve superior anti-cancer efficacy.
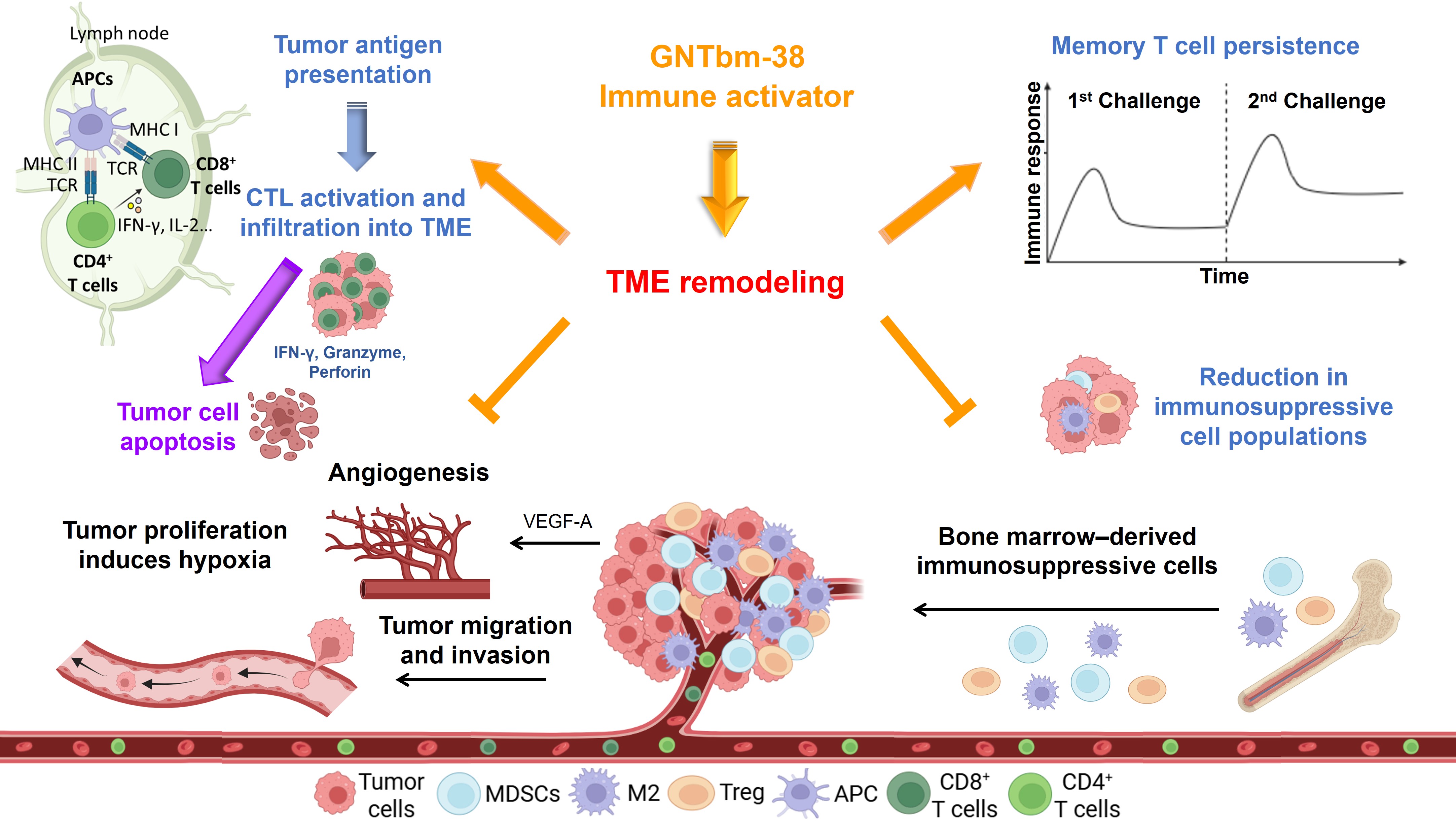
Leveraging a proprietary de novo design platform, GNTbm has successfully developed a next-generation epigenetic immunoactivator with dual-functional superiority. Through advanced structural optimization and computational modeling, GNTbm has significantly enhanced both epigenetic potency and immune activation capability, enabling deep and sustained remodeling of the tumor microenvironment (TME). GNTbm-38 was selected as the lead candidate—a novel small-molecule benzamide designed to redefine the therapeutic potential of epigenetic drugs.
In multiple immunocompetent, high tumor burden animal models, oral GNTbm-38 demonstrated exceptional immune activation. Notably, when combined with immunomodulatory multi-kinase inhibitors, GNTbm-38 achieved full immune system engagement at substantially lower doses, resulting in remarkably high objective response rates (ORR). More importantly, tumor re-challenge studies demonstrated durable immunological memory, effectively preventing tumor relapse—highlighting its potential to deliver long-term disease control.
GNTbm-38 has completed global patent filings and has been granted approvals in 42 countries to date. Preclinical studies have been completed, and IND applications were approved in the United States and China in 2026. Multinational Phase I clinical trials are scheduled to be initiated in 2026.
GNTbm-38 is one of the core orally administered cancer immunotherapy agents independently developed by GNTbm, representing a key innovation in the company’s next-generation cancer immunotherapy pipeline.
Mechanisms of GNTbm-38 in Inhibiting Tumor Growth and Remodeling the Tumor Microenvironment
4. Immunoregulating multi-kinase inhibitor screening platform
Multi-kinase inhibitors (tyrosine kinase inhibitors, TKIs) are among the most important classes of targeted anti-cancer therapies, with multiple agents successfully approved worldwide. GNTbm has identified that certain classes of TKIs not only inhibit tumor growth but also possess potent immunomodulatory activity, enabling the reprogramming of immune responses and improvement of the tumor microenvironment (TME).
When combined with GNTbm-38, these TKIs can further activate the host immune system through comprehensive TME reprogramming, leading to robust anti-tumor immune responses and the establishment of durable immunological memory. This translates into sustained tumor control with a reduced likelihood of recurrence.
Importantly, unlike conventional strategies that rely on immune checkpoint inhibitors, this novel combination approach can achieve profound TME remodeling without the need for checkpoint blockade. Through multi-dimensional mechanisms—including tumor vascular normalization, alleviation of hypoxia, reduction of lactate accumulation, enhanced infiltration of cytotoxic T lymphocytes (CTLs), suppression of immunosuppressive cell populations, and regulation of cytokine and chemokine networks—this strategy enables a comprehensive reactivation of anti-tumor immunity. GNTbm’s core therapeutic paradigm is defined as “GNTbm-38 + Y,” where Y represents a selected TKI, with the proprietary GNTbm-TKI expected to deliver optimal therapeutic synergy.
Building on years of research, GNTbm has successfully developed GNTbm-TKI, a structurally novel multi-kinase inhibitor targeting key oncogenic and immunoregulatory pathways, including TYRO3, AXL, c-MER, BTK, ROS1, NTRK2, MET, and VEGFR2. This broad targeting profile underpins its strong immunomodulatory potential. In preclinical studies, the combination of GNTbm-TKI and GNTbm-38 achieved objective response rates (ORR) exceeding 80%. Notably, treated models demonstrated durable immune memory, with rapid tumor clearance upon re-challenge, indicating long-term tumor suppression and prevention of relapse. Ongoing studies continue to further validate these findings.
In parallel, GNTbm-TKI has completed patent filings in 53 countries and is currently in preclinical development. IND submissions in the United States, Taiwan, and China are planned by the end of 2026, advancing toward clinical-stage development.
The mechanisms of GNTbm-TKI in inhibiting cancer cell growth/invasion and metastasis/tumor angiogenesis and remodeling tumor microenvironment

.jpg)