Epigenetic Immunoactivator
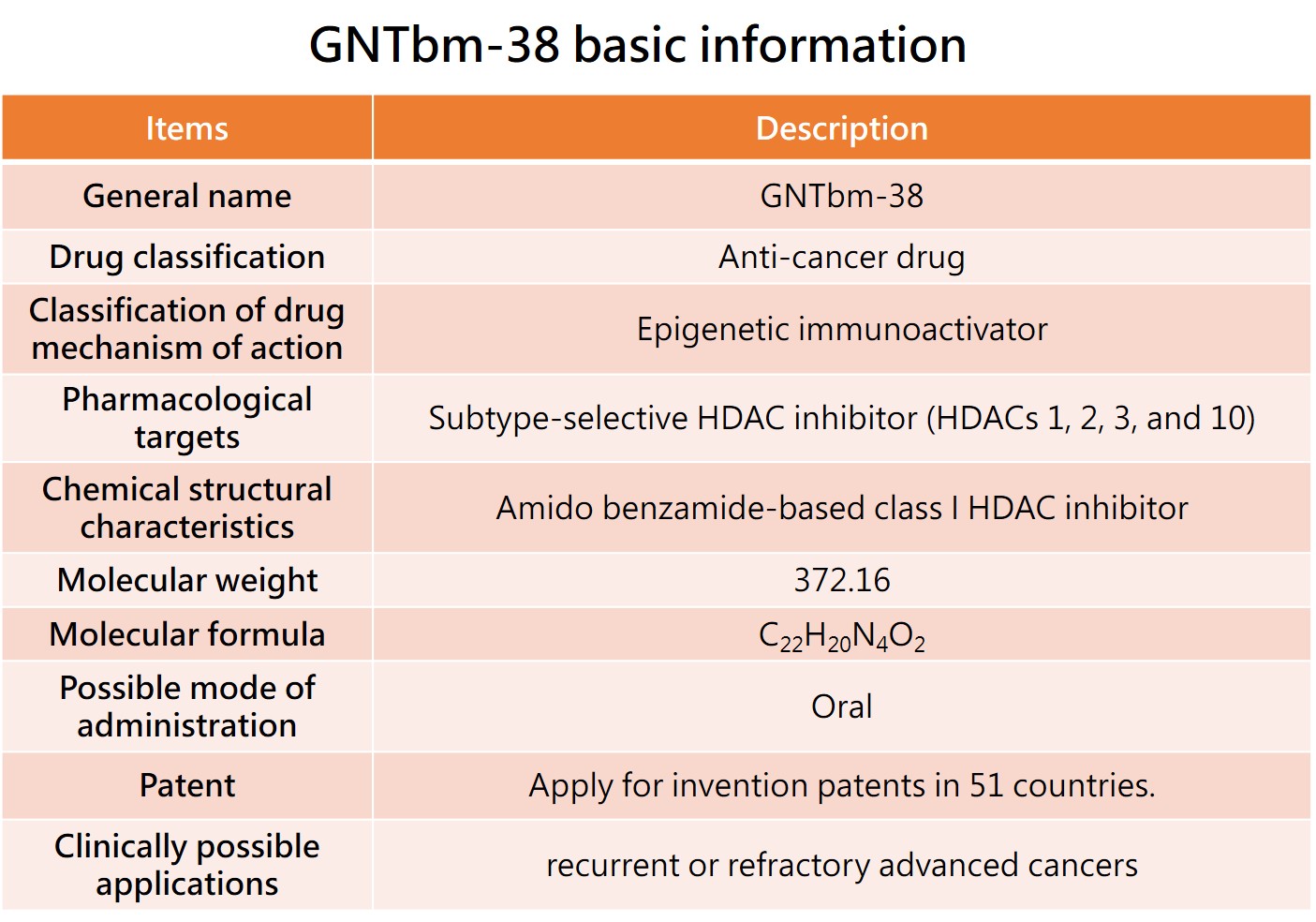
The GNTbm R&D team has independently developed a new structural epigenetic immunoactivator, named GNTbm-38, after years of R&D. It is a dual-function drug that has been screened for innovative, small-molecule, oral, and excellent epigenetic regulation and immune modulation activities. Studies on enzyme, cell, and animal test platforms have shown that GNTbm-38 has excellent epigenetic and immune modulation activities. When used in combination with TKIs that inhibit specific targets, it demonstrates very superior activity in remodeling the TME, significantly increasing the tumor response rate. GNTbm-38 can be used as a monotherapy or in combination with TKIs that inhibit specific targets for cancer immunotherapy. The chemical structure of GNTbm-38 falls under the category of benzamide-based, serving as a selective class I HDAC inhibitor. GNTbm has obtained multiple invention patents, including in the United States. GNTbm-38 is one of the essential components in cancer combination immunotherapy.
GNTbm-38 has completed preclinical studies and received FDA clearance in February 2026 to initiate a Phase 1 clinical trial, followed by a Phase 2 clinical trial, with R/R PTCL as the first indication.
An application for orphan drug designation in the United States will be submitted. Once the R/R PTCL drug approval is obtained, new indications will be expanded, by using 'GNTbm-38 + Z’, where Z could be a TKI that inhibits unique targets or a anti-PD-1/VEGF bispecific antibody, for treatment of various advanced solid tumors.
1. The first indication of GNTbm-38---R/R PTCL
Over the past several years, research has established that relapsed/refractory peripheral T-cell lymphoma (R/R PTCL) is highly aggressive and exhibits significant disease heterogeneity, with pathogenesis closely linked to dysregulation of epigenetic control. Consequently, multiple HDAC inhibitors have entered clinical development and received approval for the treatment of R/R PTCL, including Belinostat, Romidepsin, and Tucidinostat.
Compared with B-cell lymphomas, T-cell lymphomas present greater therapeutic challenges due to the absence of standard treatment regimens, diverse subtypes, and high heterogeneity, resulting in poor overall prognosis and a substantial unmet medical need in clinical practice.
In preclinical studies, GNTbm-38 demonstrated significant antitumor activity in a murine R/R angioimmunoblastic T-cell lymphoma (AITL) PDX model. Compared with the positive control Belinostat administered intravenously, GNTbm-38 delivered orally exhibited superior antitumor efficacy, further supporting its potential as a monotherapy for R/R PTCL.
Clinical Development Strategy:
Phase Ia Trial: Enrollment of patients with advanced solid tumors for a dose-escalation study to evaluate dose-limiting toxicity (DLT), maximum tolerated dose (MTD), and recommended Phase II dose (RP2D).
Phase Ib Trial: Multi-countries enrollment of patients with R/R PTCL to further assess efficacy and safety.
If clinical results are favorable, the data from these trials will be used to support applications for orphan drug designation (ODD) in the United States, European Union, Japan, and China.
Following the attainment of multinational orphan drug status, GNTbm-38 will pursue Fast Track designation or similar accelerated regulatory pathways, and a pivotal Phase II study will be conducted with synchronized patient enrollment in the United States, Canada, Australia, Europe, Japan, Taiwan, and China.
If the pivotal trial demonstrates satisfactory efficacy and safety, the results will form the basis for new drug application (NDA) submissions to regulatory authorities in multiple countries, aiming to provide patients with R/R PTCL a novel therapeutic option.
2. New Indication for GNTbm-38 — MYC/BCL2 Double-Expressor Diffuse Large B-Cell Lymphoma (DE-DLBCL)
Diffuse large B-cell lymphoma (DLBCL) has been treated primarily with the standard R-CHOP regimen as first-line therapy for more than 20 years, achieving complete remission (CR) rates of approximately 60–70%. However, recent studies have highlighted the substantial heterogeneity of DLBCL, which is closely associated with dysregulation of epigenetic control, further impacting treatment response and prognosis.
Among DLBCL patients, high-grade B-cell lymphoma harboring rearrangements in MYC, BCL2, and/or BCL6 genes (double-hit or triple-hit lymphoma) is associated with particularly poor outcomes, though such cases account for only 5–8% of patients. In contrast, MYC/BCL2 protein-high DLBCL (MYC/BCL2 DE-DLBCL) represents approximately 30% of cases, constituting a clinically significant patient population. Although the World Health Organization (WHO) has not designated this as a distinct subtype, patients with MYC/BCL2 DE-DLBCL treated with R-CHOP exhibit lower CR rates, as well as significantly reduced 5-year progression-free survival (PFS) and overall survival (OS) compared with patients lacking high protein expression, underscoring the substantial unmet medical need in this population.
In 2024, Tucidinostat in combination with R-CHOP was shown to significantly improve first-line CR rates in patients with MYC/BCL2 DE-DLBCL (from 61.8% to 73%), with event-free survival (EFS) reaching statistical significance. This combination has also received approval in China as a new indication, validating the clinical feasibility of targeting epigenetic regulation as a therapeutic strategy.
Building on this foundation, GNTbm-38 is being developed with differentiated advantages. As a selective class I HDAC inhibitor, GNTbm-38 not only exhibits enhanced antitumor activity but also possesses immunomodulatory properties, with the potential to simultaneously regulate tumor cell proliferation and the tumor immune microenvironment. Compared with Tucidinostat, GNTbm-38 demonstrates stronger epigenetic reprogramming capabilities and the potential for deeper treatment responses, with a target CR rate of 85%.
According to WHO criteria, MYC protein expression above 40% and BCL2 expression above 50% define the high-expression population. High MYC expression promotes tumor cell proliferation, while high BCL2 expression inhibits apoptosis, collectively conferring resistance to R-CHOP. Achieving complete remission is therefore critical to prolonging PFS and OS.
Based on these mechanisms and clinical needs, a novel stratified treatment strategy has been proposed, positioning GNTbm-38 as the backbone therapy in combination with a BTK inhibitor and Rituximab:
Early CR: Patients achieving CR after the first two treatment cycles of GNTbm-38 + BTK inhibitor + Rituximab continue this combination for six cycles to minimize chemotherapy exposure.Partial Response or Stable Disease: Patients achieving only partial response (PR) or stable disease (SD) proceed to receive six cycles of CHOP in addition to the combination therapy to achieve CR.
This adaptive strategy allows treatment to be tailored based on early response, maximizing efficacy while managing toxicity. It also harnesses GNTbm-38’s potential to improve CR rates, overcome drug resistance, and serve as a versatile platform for combination therapy, offering a potentially transformative treatment option for patients with MYC/BCL2 DE-DLBCL.
3. New indication of GNTbm-38 — microsatellite stable/metastatic colorectal cancer (MSS/pMMR mCRC)
Colorectal cancer (CRC) is one of the leading causes of cancer-related mortality worldwide, with approximately 2 million new cases and 1.1 million deaths each year, representing a significant unmet clinical need. Current cancer immunotherapies are largely effective only in patients with high microsatellite instability (MSI-H); however, this population accounts for only about 5% of CRC cases. The majority of patients (~90%) are microsatellite stable (MSS/pMMR) and exhibit poor response to immunotherapy, representing a classic "immune-cold tumor" scenario.
For unresectable advanced or metastatic colorectal cancer (mCRC), treatment is primarily systemic, but MSS/pMMR patients lack effective immunotherapy options. A breakthrough emerged in 2024: a triplet regimen of Tucidinostat combined with an anti-PD-1 antibody and Bevacizumab. This combination synergistically remodels the tumor microenvironment through epigenetic modulation and anti-angiogenic effects, converting MSS mCRC from an immune-cold to an immune-hot tumor. The regimen achieved an objective response rate (ORR) of 44% and a median progression-free survival (mPFS) of 7.3 months, representing a historic milestone.
Currently, this triplet therapy has progressed to pivotal Phase III clinical trials, with Fruquintinib as the control and overall survival (OS) as the primary endpoint, with the potential to yield positive results that further validate the clinical value of the "epigenetic × immune × anti-angiogenesis" strategy.
Building on this foundation, GNTbm-38, a next-generation epigenetic modulator, demonstrates superior tumor immune activation and epigenetic reprogramming compared to Tucidinostat, with the potential to further enhance immunotherapy response rates. GNTbm-38 will employ an innovative combination strategy with an anti-PD-1/VEGF bispecific antibody, simultaneously achieving immune checkpoint blockade and tumor vascular modulation, with the goal of improving efficacy while reducing the toxicity associated with multi-drug regimens.
Clinically, GNTbm-38 will focus on third-line treatment of advanced colorectal cancer, specifically targeting MSS/pMMR patients for proof-of-concept (POC) studies. Through its unique immune activation mechanism, GNTbm-38 can serve as a backbone therapy in combination regimens, synergizing with bispecific antibodies to further strengthen tumor microenvironment remodeling.
This strategy not only has the potential to overcome the long-standing immunotherapy resistance in MSS CRC but also represents a key clinical validation of GNTbm-38’s capability to convert "cold tumors" into "hot tumors" in advanced solid tumors, highlighting its core value as a platform immune-modulatory agent.