Tucidinostat/Chidamide (Trade name in Mandarin :剋必達®, Trade name in English : Kepida®)
Tucidinostat/Chidamide (Trade name in Mandarin :剋必達®, Trade name in English : Kepida®)
Tucidinostat is a globally recognized novel therapeutic currently being developed by four pharmaceutical companies across various regions. GNTbm secured the exclusive rights for the research, development, manufacturing, and commercialization of Tucidinostat in Taiwan in September 2013. As one of the most promising candidates in the global landscape of HDAC inhibitors, Tucidinostat functions as a subtype-selective histone deacetylase inhibitor (also known as an epigenetic modulator) that specifically targets HDAC isoforms 1, 2, 3, and 10. By blocking the activity of histone deacetylases, the drug facilitates the transition of tightly wound chromatin into a more relaxed structure, thereby reactivating tumor suppressor genes or immune antigen genes previously silenced by cancer cells. Acting as a genomic switch in anti-cancer strategies, Tucidinostat exposes disguised malignant cells to the immune system. It serves as a pivotal epigenetic agent in the conversion of immunologically cold tumors into hot tumors.
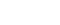
Tucidinostat has been approved for four indications globally, with two additional pivotal Phase III clinical trials—leveraging tumor immunotherapy-based mechanisms of action—approaching completion. To date, among histone deacetylase (HDAC) inhibitors in global development, Tucidinostat stands out as one of the agents with the highest number of regulatory approvals and the broadest range of indications. Its clinical applications have expanded from hematologic malignancies to multiple solid tumors, demonstrating a clear advantage in global clinical development.
In China, Tucidinostat has been approved for three indications: relapsed or refractory peripheral T-cell lymphoma (R/R PTCL), HR+/HER-2− advanced breast cancer, and MYC/BCL2 double-expressor diffuse large B-cell lymphoma (DLBCL).
In Japan, Tucidinostat has also been approved for relapsed or refractory adult T-cell leukemia/lymphoma (ATL) and relapsed or refractory peripheral T-cell lymphoma.
In Western markets, a global pivotal Phase III trial evaluating Tucidinostat in combination with Opdivo as a first-line treatment for advanced melanoma has completed patient enrollment across 17 countries, highlighting its strong potential in immunotherapy-based combination strategies.
In Taiwan, Tucidinostat has obtained approval as a new chemical entity (NCE) for the treatment of HR+/HER-2− advanced breast cancer and has been reimbursed under the National Health Insurance (NHI) program since December 1, 2025. Ongoing efforts are focused on expanding its indications, including relapsed or refractory peripheral T-cell lymphoma.
Basic Information of Tucidinostat/Kepida®
.png)
Tucidinostat Patent Licensing and Development
After the originator Chipscreen Biosciences completed the global multinational patent layout of Tucidinostat, it successively licensed the patents to many international new drug development companies and well-known big pharmaceutical companies, and the international licensing and development status is as follows:
In 2006, Chipscreen Biosciences has licensed the development and commercialization rights of Tucidinostate to HUYABIO International, LLC for all global markets excluding China, Hong Kong, and Taiwan.
In 2013, Chipscreen Biosciences licensed the Taiwan rights to GNTbm.
In 2020, HUYABIO International sublicensed the rights for eight countries, including Japan, South Korea, and Southeast Asian nations, to Japan’s Meiji Seika Pharma.
Tucidinostat is currently developed globally and owned by four pharmaceutical companies, cooperating with each other.
.jpg)
Table 1. The current status of Tucidinostat clinical development.