Tucidinostat/Chidamide (Trade name in Mandarin :剋必達®, Trade name in English : Kepida®)
Tucidinostat's first indication in Taiwan – combined with Exemestane for the treatment of HR+/Her-2-advanced breast cancer
1. Kepida® has obtained the drug license for domestically developed new new drug after 10 years of development in Taiwan
The trade name of Tucidinostat in Taiwan is Kepida®, a new generation oral epigenetic modulator. It is a subtype-selective HDAC inhibitor, and GNTbm spent 10 years and invested hundreds of millions in research and development to obtain the domestic certification for a new drug.

2. Tucidinostat meets the medical needs of HR+/Her-2- advanced breast cancer patients as a second-line treatment
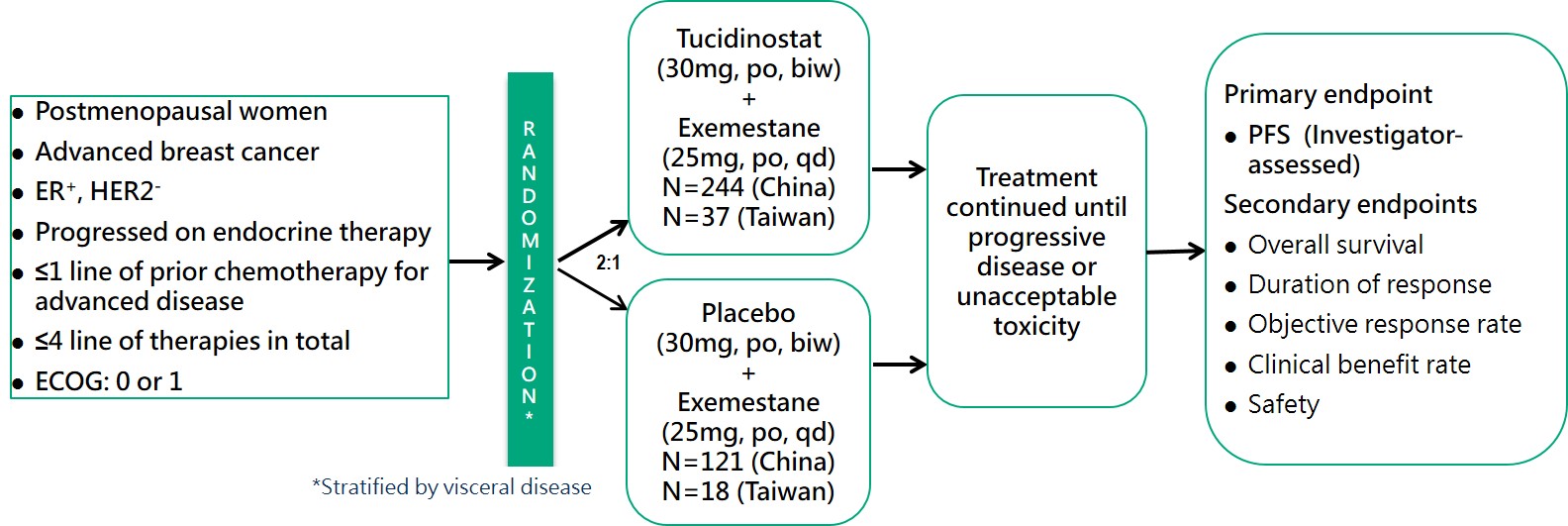
According to the NCCN Guidelines and the consensus of the Taiwan Breast Cancer Society (TBCS), the primary recommendation for first-line treatment of HR+/HER-2- advanced breast cancer is endocrine therapy in combination with CDK4/6 inhibitors. Under Taiwan’s National Health Insurance (NHI) regulations, patients are eligible for a lifetime maximum of 24 months of reimbursed CDK4/6 inhibitor treatment. International literature further supports this standard, reporting exceptional therapeutic efficacy with a median Progression-Free Survival (mPFS) of approximately 24 months. These factors collectively indicate that roughly 70% of HR+/HER-2- advanced breast cancer patients in Taiwan receive frontline endocrine therapy combined with CDK4/6 inhibitors.
Following the failure of first-line treatment, patients frequently develop dual resistance to both endocrine therapy and CDK4/6 inhibitors, significantly limiting subsequent second-line options. The NCCN Guidelines offer restricted recommendations for this setting:
1. For patients who have not previously received a CDK4/6 inhibitor, a combination of a CDK4/6 inhibitor with endocrine therapy is recommended.
2. For patients with PIK3CA mutations, Alpelisib (a PI3K inhibitor) combined with Fulvestrant is recommended.
3. Alternatively, Everolimus (an mTOR inhibitor) combined with Exemestane may be used.
Tucidinostat (an HDAC inhibitor) combined with Exemestane represents an alternative strategy for advanced breast cancer. As an epigenetic modulator, it regulates gene expression through a mechanism distinct from endocrine therapy and CDK4/6 inhibitors, with potential to overcome treatment resistance.
GNTbm plans to conduct a real-world study in Taiwan enrolling HR+/HER2− advanced breast cancer patients who progressed after first-line endocrine therapy plus a CDK4/6 inhibitor. Patients will receive Tucidinostat with Exemestane as second-line treatment to evaluate its clinical efficacy.
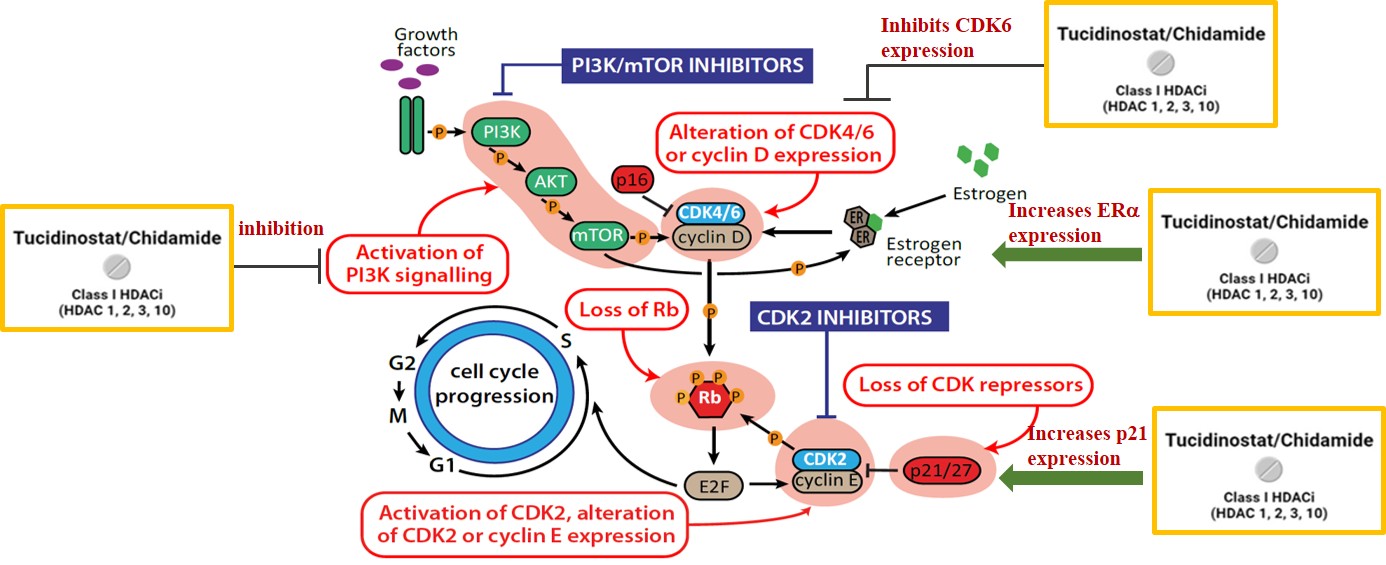
(1) Providing new treatment options: Tucidinostat is an epigenetic modulator regulating gene expression, the only multi-target agent that has been recommended as the first choice for second-line treatment by the expert consensus of the Taiwan Society of Breast Medicine (Grade A).
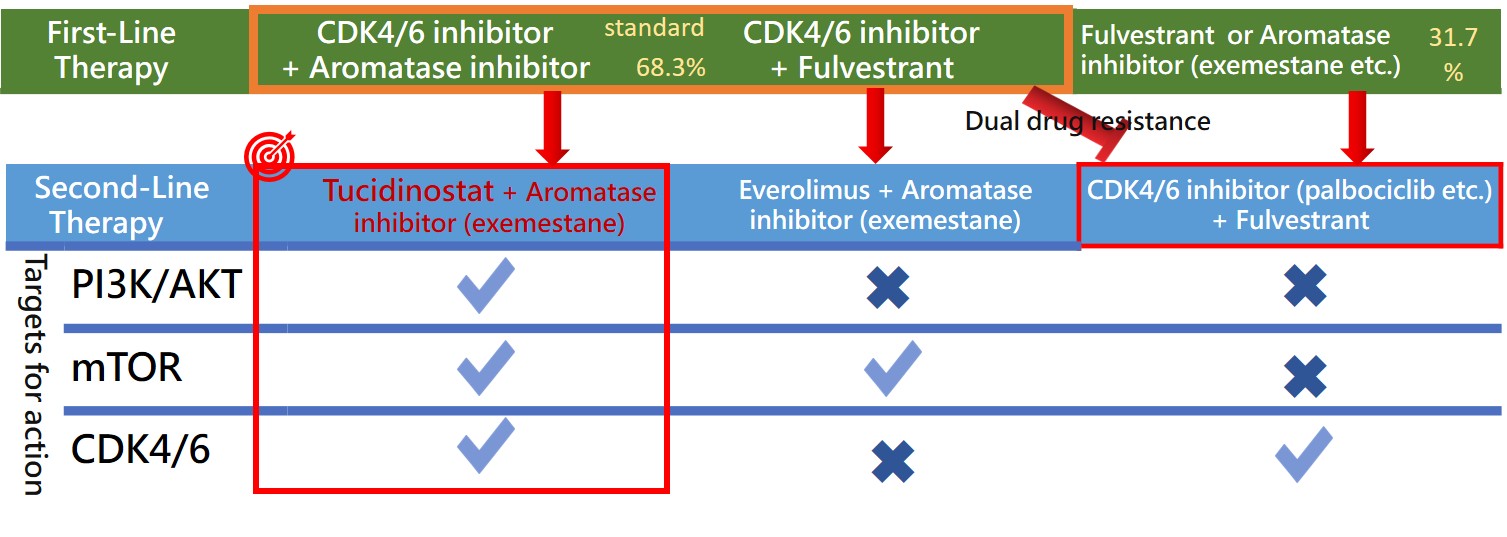
(2) Clinical efficacy comparison: Kepida® has demonstrated clinical efficacy comparable to CDK4/6 inhibitors.
3. Pivotal Phase III Clinical Trial for the Tucidinostat Treatment of HR+/HER-2- Advanced Breast Cancer (CDM301)
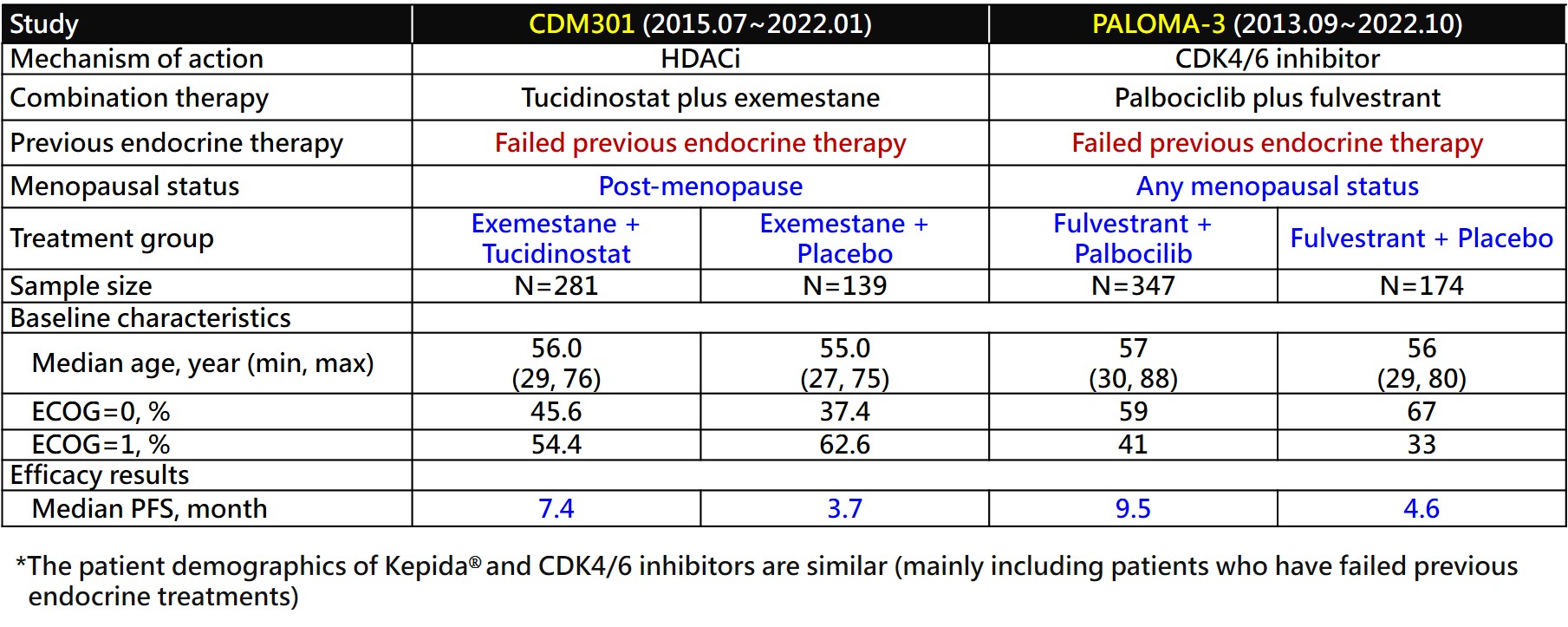
GNTbm submitted the clinical trial protocol for Phase III to treat advanced breast cancer to the TFDA/CDE for review and received recommendations from TFDA/CDE. The trial design and inclusion criteria for patients were the same as that of the clinical trial conducted in China, and it was suggested that the clinical data from Taiwan were combined with the clinical data from China to serve as the endpoints for this trial.
The clinical trial, called CDM301, recruited patients with HR+/Her-2- advanced breast cancer who had failed or relapsed with at least one previous endocrine therapy, treated with Tucidinostat plus Exemestane. The patients were stratified according to presence or absence of visceral metastasis, and randomly assigned 2:1 to the experimental group and the placebo group for treatment until disease progression or withdrawal from the trial due to drug toxicity, and the primary endpoint was PFS assessed by clinical investigators. The trial period was about 5 years.
(1) Efficacy assessment
CDM301 is a well-designed, placebo-controlled, randomized, multicenter, pivotal Phase III clinical trial in Taiwan and China. A total of 420 patients were enrolled and randomized 2:1 to Tucidinostat plus Exemestane (experimental group) and placebo plus Exemestane (placebo group), with 281 and 139 patients, respectively. The efficacy outcomes mPFS of the primary endpoint of the trial were 7.4 months and 3.7 months (HR=0.716; 95% CI, 0.562 ~ 0.911; P=0.0066) for the experimental and placebo groups, respectively. Data analysis of 55 patients in Taiwan showed that Tucidinostat combined with Exemestane (experimental group) and placebo plus Exemestane (placebo group) were randomly assigned 2:1, with 37 and 18 cases, respectively. The mPFS of the primary endpoint were 8.6 months and 3.7 months (HR=0.516; 95% CI, 0.268 ~ 0.993; P=0.0477), for the experimental and placebo groups, respectively. In 420 patients, the objective response rate (ORR) of the secondary endpoint efficacy of the trial was 16.73% in the experimental group and 7.91% in the placebo group, which showed a statistically significant difference.
(2) Safety data
Common adverse events in the Tucidinostat combined with Exemestane groups were hematologic toxicity and gastrointestinal toxicity, both of which were controllable adverse reactions that could be restored by reducing the dose of investigational drug or suspending the investigational drug.