Tucidinostat/Chidamide (Trade name in Mandarin :剋必達®, Trade name in English : Kepida®)
Tucidinostat/Chidamide (Trade name in Mandarin :剋必達®, Trade name in English : Kepida®)
Tucidinostat is a new small molecule drug developed worldwide by four pharmaceutical companies across different countries. GNTbm obtained the rights for the research & development, manufacturing, and sales of Tucidinostat in Taiwan in September 2013. Tucidinostat is one of the most promising histone deacetylase inhibitors under global development and is classified as a subtype-selective HDAC inhibitor, also known as an epigenetic modulator. It exerts its effects by selectively inhibiting HDACs 1, 2, 3, and 10, making it a very popular new targeted anti-tumor drug.
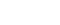
Tucidinostat has been approved for four indications globally, and two pivotal Phase III clinical trials for cancer immunotherapy are about to be completed. So far, in the global development of HDAC inhibitors, Tucidinostat is the most approved HDACi for cancer indications, expanding from lymphoma to solid tumors.
Tucidinostat is being actively developed in Taiwan and also by the other three companies around the world for different new indications. Firstly in China, Tucidinostat has obtained drug approvals for three new indications: treatment of relapsed or refractory peripheral T-cell lymphoma (R/R PTCL), HR+/Her-2- advanced breast cancer, and MYC/BCL2 double-expressing DLBCL; in Japan, Tucidinostat has also obtained drug approvals for two new indications: relapsed or refractory adult T-cell lymphoma (R/R ATL) and relapsed or refractory peripheral T-cell lymphoma (R/T PTCL); in the United States, the European Union, and other countries, a pivotal Phase III trial of Tucidinostat combined with Opdivo® for first-line treatment of advanced melanoma has completed patient enrollment in 17 countries worldwide; and in Taiwan, Tucidinostat has obtained the first drug approval for the treatment of HR+/Her-2- advanced breast cancer, and is expanding the second indication for the treatment of relapsed or refractory peripheral T-cell lymphoma (R/R PTCL).
Basic Information of Tucidinostat/Kepida®
.png)
Tucidinostat Patent Licensing and Development
After the originator Chipscreen Biosciences completed the global multinational patent layout of Tucidinostat, it successively licensed the patents to many international new drug development companies and well-known big pharmaceutical companies, and the international licensing and development status is as follows:
In 2006, Chipscreen Biosciences granted patents outside China, Hong Kong and Taiwan to HUYABIO International, LLC.
In 2013, Chipscreen Biosciences authorized Taiwan right to GNTbm.
In 2020, HUYABIO International reauthorizes a total of eight countries, including Japan, Korea and Southeast Asia to Meiji Seika Pharma of Japan.
Tucidinostat is currently developed globally and owned by four pharmaceutical companies, cooperating with each other.
.jpg)
Table 1. The current status of Tucidinostat clinical development.